Its name sounds like something out of science fiction, but it’s just science. The cyclotron, located in the depths of the Centennial building at the QEII’s VG site, hums with energy day and night.
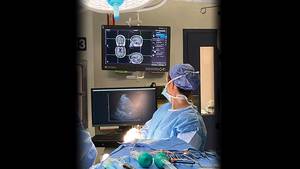
The cyclotron is a device that creates medical isotopes. A medical isotope is a very small amount of radioactive material used in the diagnosis of cancer and other diseases. These isotopes are used every day in the QEII’s positron emission tomography-computed tomography (PET CT) scanner.
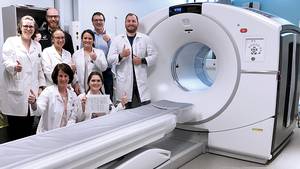
The production of isotopes is no easy process and is managed by a very dedicated team at the QEII.
Antoun Bou Laouz, Melissa Sponagle, Julian Walcott and Karen Aucoin produce and package the medical isotope known as fluorodeoxyglucose (FDG) for transport to the PET CT scanner upstairs. This small quantity of radioactive material decays quickly however, meaning the time spent from production to medical use is limited, making every second critical.
Not only is the team under tight deadlines daily, they also have to be aware of the constant presence of radioactive materials. Safety is a top priority and the team goes the extra mile to ensure patients and staff are protected.
The cyclotron is self-shielded consisting of eight tanks filled with boronated water and lead. The shield has two doors, weighing an incredible 14 tons.
“We also have radiation detectors in the ceiling. They constantly collect data, so we can monitor the radiation level in here at all times,” says Melissa.
The team test and check safety parameters around the cyclotron every month and if anything is out of place, the cyclotron will not run. They also have to pay particular attention to environmental conditions in the room, such as humidity in the summer when they run several large dehumidifiers in the area to avoid moisture build-up.
The final product of the cyclotron is placed in a glass vial, and then placed in a heavy, tungsten shield pot and then into another shielded box for transport. Before it leaves, the team checks the container for radiation and makes sure it’s safe for transport.
Before the cyclotron at the QEII
When PET emerged in the 1990s as an effective tool for assessing and diagnosing cancer, many hospitals in Canada wanted this new technology. However, as with many new medical advances, the equipment was expensive and governments alone were wary of having to fund it across Canada.
Before the QEII had its own cyclotron, the PET CT’s supply of FDG isotopes had to be shipped in from Montreal every day to service an ever-increasing demand for the scans in Atlantic Canada. It proved to be expensive and sometimes ineffective. “We were very dependent on the transportation system,” says Dr. Andrew Ross of Diagnostic Radiology.
Because FDG decays very quickly, shipping it to Halifax from Montreal limited the effectiveness of the product by the time it was used in Halifax, which impacted wait times.
“In order to get 300 millicuries [unit of radioactivity] here for use, they were shipping 1,200 to 1,300 millicuries out of the cyclotron in Montreal to get that 300 here for the day,” says Dr. Ross.
For those patients who could afford to go to Quebec, the province picked up the tab for the test, but the transport was up to the patient. The process was not ideal, which is why the team at the QEII decided it was time to make their own isotopes here in Nova Scotia. This process was in place for two years, until the cyclotron came online in 2009.
The PET CT scanner and cyclotron project was made the flagship project of a major fundraising campaign and received more than $3-million from the QEII’s radiologists, Bell Aliant, the provincial government and the QEII Foundation.
“Having a local source makes it so much easier,” says Dr. Ross. "By running for an hour in the morning, they can easily get the 300 millicuries here for the day.”
Dr. Ross says that with the cyclotron running, they can complete 12 to 13 scans every day. This is compared to just eight or nine per day when they received the isotopes from Montreal. In addition to seeing more patients, it is also a more reliable service with the ability to test extra patients when necessary.
For Nova Scotia and other Atlantic provinces, this couldn’t be more important.
What's next?
The QEII’s PET CT scanner and cyclotron are the only ones in Nova Scotia and although FDG is produced all over Canada, whenever a new facility enters production, Health Canada treats it as a new drug.
“We are working on getting a licence from Health Canada,” says Brian Martell, manager special imaging at the QEII. “We are currently operating under a CTA (clinical trial application); we want to be able to operate as a manufacturer. It’s very stringent. They hold us to the same standard that they hold the drug companies to.
“One of the long term visions is that if we had the licence we would be able to supply the product to other provinces.”